 A bar of soap is a chemical miracle – a little scented block that unleashes molecular forces when dunked in water. So how does this little miracle, that saves lives daily by sanitizing the many deadly germs clinging to us, actually work ?
A bar of soap is a chemical miracle – a little scented block that unleashes molecular forces when dunked in water. So how does this little miracle, that saves lives daily by sanitizing the many deadly germs clinging to us, actually work ?
HOW
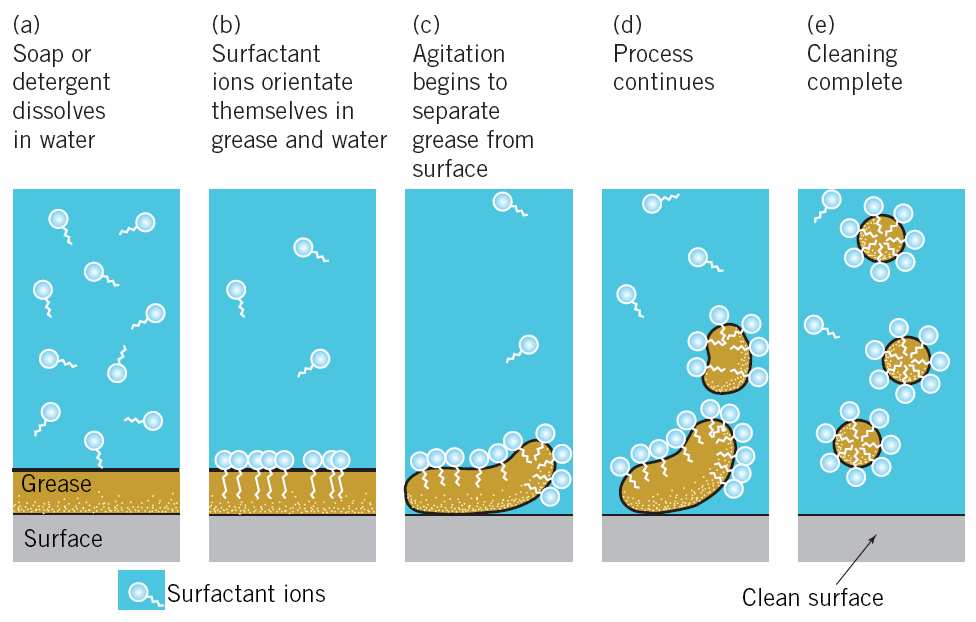
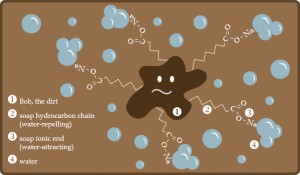
Well, soap molecules have a rather peculiar property of having one end attracted by water and the other repelled by it. This gives soap two key properties for getting things clean :
First : It reduces the attraction that water molecules have for each other, compelling them to spread themselves more effectively over whatever has been put in the soapy water;
Second : It allows the soap molecules to work their way under dirt and prise it off, floating it away surrounded by bundles of molecules – which also stops the dirt simply floating back on to the clothes again.
A (BRIEF) HISTORY OF SOAP
Oddly enough, although the Babylonians first made soap (presumably by accident) around 4,800 years ago, it was primarily used to treat skin conditions, its dirt-shifting powers weren’t recognized until medieval times.
Given that so many bacterial and viral infections (especially colds) are transmitted by skin contact, the invention of soap must rate as one of the major medical breakthroughs in history.